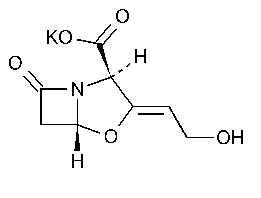
Clavulanate Potassium
4-Oxa-1-azabicyclo[3.2.0]heptane-2-carboxylic acid,3-(2-hydroxyethylidene)-7-oxo-,monopotassium salt,2R-(2a,3Z,5a)-.
Potassium (Z)-(2R,5R)-3-(2-hydroxyethylidene)-7-oxo-4-oxa-1-azabicyclo[3.2.0]heptane-2-carboxylate [61177-45-5].
»Clavulanate Potassium contains the equivalent of not less than 75.5percent and not more than 92.0percent of clavulanic acid (C8H9NO5),calculated on the anhydrous basis.
Packaging and storage—
Preserve in tight containers.
Labeling—
Where it is intended for use in preparing injectable dosage forms,the label states that it is sterile or must be subjected to further processing during the preparation of injectable dosage forms.
USP Reference standards á11ñ—
USP Clavulanate Lithium RS.USP Clavam-2-Carboxylate Potassium RS.USP Endotoxin RS.
Identification—
A:
The retention time of the major peak for clavulanic acid in the chromatogram of the Assay preparationcorresponds to that in the chromatogram of the Standard preparation,as obtained in the Assay.
B:
Asolution of it responds to the tests for Potassium á191ñ.
Bacterial endotoxins á85ñ—
Where the label states that Clavulanate Potassium is sterile or must be subjected to further processing during the preparation of injectable dosage forms,it contains not more than 0.03USP Endotoxin Unit per mg.
Sterility á71ñ—
Where the label states that Clavulanate Potassium is sterile,it meets the requirements when tested as directed for Membrane Filtration under Test for Sterility of the Product to be Examined.
pHá791ñ:
between 5.5and 8.0,in a solution (1in 100).
Water,Method Iá921ñ:
not more than 1.5%.
Limit of clavam-2-carboxylate potassium—
Mobile phase—
Prepare 0.1Mmonobasic sodium phosphate,adjust with phosphoric acid to a pHof 4.0±0.1,and pass through a membrane filter having a 0.5-µm or finer porosity.Make adjustments if necessary (seeSystem SuitabilityunderChromatography á621ñ).
Standard solution—
Dissolve USP Clavam-2-Carboxylate Potassium RSin water to obtain a solution having a known concentration of about 5µg per mL.
Test solution—
Transfer about 100mg of Clavulanate Potassium,accurately weighed,to a 10-mLvolumetric flask,dissolve in and dilute with water to volume,and mix.
Chromatographic system(see Chromatography á621ñ)—
The liquid chromatograph is equipped with a 210-nm detector and a 4-mm ×30-cm column that contains 3-to 10-µm packing L1.The flow rate is about 0.5mLper minute.Chromatograph theStandard solution,and record the peak responses as directed forProcedure:the column efficiency is not less than 4000theoretical plates;the tailing factor is not more than 1.5;and the relative standard deviation for replicate injections is not more than 5%.
Procedure—
Separately inject equal volumes (about 20µL)of theStandard solution and theTest solution into the chromatograph,record the chromatograms,and measure the responses for the major peaks.The relative retention times are about 0.7for clavam-2-carboxylic acid and 1.0for clavulanic acid.Calculate the percentage of clavam-2-carboxylate potassium in the specimen taken by the formula:
(C/W)(rU/rS),
in whichCis the concentration,in µg per mL,of clavam-2-carboxylate potassium in theStandard solution;Wis the quantity,in mg,of Clavulanate Potassium taken to prepare theTest solution;andrUandrSare the clavam-2-carboxylic acid peak responses obtained from theTest solutionand theStandard solution,respectively:not more than 0.01%is found.
Limit of aliphatic amines—
Internal standard solution—
Dissolve 50µLof 3-methyl-2-pentanone in water,dilute with water to 100.0mL,and mix.
Test solution—
Transfer 1.0g of Clavulanate Potassium to a centrifuge tube,add 5.0mLof Internal standard solution,5.0mLof 2Nsodium hydroxide,5.0mLof isopropyl alcohol,and 5g of sodium chloride.Shake for 1minute,and centrifuge to separate the layers.Use the upper layer.
Reference solution—
Dissolve 80.0mg of each of the following amines in 2Nhydrochloric acid:1,1-dimethylethylamine,diethylamine,tetramethylethylenediamine,1,1,3,3-tetramethylbutylamine,and N,N¢-diisopropylethylenediamine.Dilute with 2Nhydrochloric acid to 200.0mL,and mix.Transfer 5.0mLof this solution to a centrifuge tube.Add 5.0mLof Internal standard solution,10.0mLof 2Nsodium hydroxide,5.0mLof isopropyl alcohol,and 5g of sodium chloride.Shake for 1minute,and centrifuge to separate the layers.Use the upper layer.
Chromatographic system (see Chromatography á621ñ)—
The gas chromatograph is equipped with a flame-ionization detector and a 0.53-mm ×50-m capillary fused silica column that contains a 5-µm film coating of stationary phase G41.The temperatures of the column,the injection port,and the detector block are programmed as follows.
The carrier gas is helium flowing at a constant rate of about 8mLper minute.The split ratio is 1:10.Chromatograph the Reference solution,and record the peak responses as directed for Procedure:the relative retention times are about 0.55for 1,1-dimethylethylamine,0.76for diethylamine,1.0for isopropyl alcohol,1.07for tetramethylethylenediamine,1.13for 1,1,3,3-tetramethylbutylamine,1.33for N,N¢-diisopropylethylenediamine,and 1.57for bis(2-methylamino)ethyl ether.
| Time (minutes) |
Tempera- ture ( |
Elution | |
| Column | 0–7 | 35 | isothermal |
| 7–10.8 | 35–50 | linear gradient | |
| 10.8–25.8 | 150 | isothermal | |
| Injection port | 200 | ||
| Detector block | 250 |
Procedure—
Separately inject equal volumes (about 1µL)of the Test solutionand the Reference solutioninto the chromatograph,record the chromatograms,and measure the peak area responses.Calculate the percentage of each impurity in the Clavulanate Potassium taken by the formula:
0.2(ri/rS),
in which riis the peak response for an individual impurity observed in the chromatogram obtained from the Test solution;and rSis the response for the peak corresponding to the relevant analyte in the chromatogram obtained from the Reference solution.Calculate the percentage of any individual impurity for which no relevant reference compound is provided in the Reference solutionby the same formula,except for rSuse the peak response corresponding to the 1,1-dimethylethylamine peak.The total of all aliphatic amines is not more than 0.2%.
Limit of 2-ethylhexanoic acid—
Internal standard solution—
Prepare a solution of 3-cyclohexylpropionic acid in cyclohexane containing 1mg per mL.
Standard solution—
Transfer about 75mg of 2-ethylhexanoic acid,accurately weighed,to a 50-mLvolumetric flask,dilute with Internal standard solutionto volume,and mix.Transfer 1.0mLof this solution to a centrifuge tube,and add 4.0mLof 4Nhydrochloric acid.Shake for 1minute,and allow the phases to separate,centrifuging if necessary.Withdraw the lower phase,and reserve the upper phase.To the lower phase add 1.0mLof Internal standard solution,and shake for 1minute.Allow the phases to separate,centrifuging if necessary.Withdraw the upper phase,and combine with the reserved upper layer.
Test solution—
Transfer about 300mg of Clavulanate Potassium,accurately weighed,to a centrifuge tube.Add 4.0mLof 4Nhydrochloric acid,and shake with two successive 1.0mLportions of Internal standard solution.Allow the phases to separate,centrifuging if necessary.Use the combined upper phases.
Chromatographic system (see Chromatography á621ñ)—
The gas chromatograph is equipped with a flame-ionization detector and a 0.53-mm ×25-m capillary fused silica column that contains a 1-µm film coating of stationary phase G35.The temperatures of the column,the injection port,and the detector block are programmed as follows.
The carrier gas is hydrogen flowing at a constant rate of about 100mLper minute.Chromatograph the Standard solution,and record the peak responses as directed for Procedure:the resolution,R,between the 2-ethylhexanoic acid peak and the 3-cyclohexylpropionic acid peak is not less than 2.0.
| Time (minutes) |
Tempera- ture ( |
Rate ( minute) |
Elution | |
| Column | 0–2 | 40 | – | isothermal |
| 2–7.3 | 40–200 | 30 | linear gradient | |
| 7.3–10.3 | 200 | – | isothermal | |
| Injection port |
200 | |||
| Detector block |
300 |
Procedure—
Separately inject equal volumes (about 1µL)of the Standard solutionand the Test solutioninto the chromatograph,record the chromatograms,and measure the peak area responses.Calculate the percentage of 2-ethylhexanoic acid in the Clavulanate Potassium taken by the formula:
2(WS/WU)(RU/RS),
in which WSis the weight,in mg,of 2-ethylhexanoic acid taken to prepare the Standard solution;WUis the weight,in mg,of Clavulanate Potassium taken to prepare the Test solution;and RUand RSare the ratios of the 2-ethylhexanoic acid peak to the 3-cyclohexylpropionic acid peak observed in the chromatograms obtained from the Test solutionand the Standard solution,respectively.Not more than 0.8%is found.
Chromatographic purity—
Solution A—
Prepare a solution of 0.05Mmonobasic sodium phosphate,adjust with phosphoric acid to a pHof 4.0±0.1,and pass through a filter having a 0.5-µm or finer porosity.
Solution B—
Prepare a mixture of Solution Aand methanol (50:50).
Mobile phase—
Use variable mixtures of Solution Aand Solution Bas directed for Chromatographic system.Make adjustments to either Solutionas necessary (see System Suitabilityunder Chromatography á621ñ).
Standard solution—
Prepare a solution of USP Clavulanate Lithium RSin Solution Ahaving a known concentration of about 0.1mg per mL.
Test solution—
Prepare a solution of Clavulanate Potassium in Solution Acontaining 10.0mg per mL.
Resolution solution—
Prepare a solution of USP Clavulanate Lithium RSand amoxicillin in Solution Acontaining 0.1mg of each per mL.
Chromatographic system(see Chromatography á621ñ)—
The liquid chromatograph is equipped with a 230-nm detector and a 4.6-mm ×10-cm column that contains 5-µm packing L1and is maintained at a constant temperature of about 40 .The flow rate is about 1mLper minute.The system is programmed to provide a mobile phase consisting of variable mixtures of Solution Aand Solution B.The system is equilibrated for 15minutes with 100%Solution A,and held at that composition for 4minutes after injection of the solution under test,after which the proportion of Solution Bis increased linearly from 0%to 50%over a period of 11minutes.The system is held at that composition for 3minutes,and is then changed to 100%Solution Afor 6minutes.Chromatograph the Resolution solution,and record the peak responses as directed for Procedure:the relative retention times are about 1.0for clavulanic acid and 2.5for amoxicillin;the tailing factor for the clavulanic acid peak is not more than 2.0;the column efficiency determined from the clavulanic acid peak is not less than 2000theoretical plates;and the resolution,R,between the clavulanic acid peak and the amoxicillin peak is not less than 13.Chromatograph the Standard solution,and record the peak responses as directed for Procedure:the relative standard deviation for replicate injections is not more than 2%.
.The flow rate is about 1mLper minute.The system is programmed to provide a mobile phase consisting of variable mixtures of Solution Aand Solution B.The system is equilibrated for 15minutes with 100%Solution A,and held at that composition for 4minutes after injection of the solution under test,after which the proportion of Solution Bis increased linearly from 0%to 50%over a period of 11minutes.The system is held at that composition for 3minutes,and is then changed to 100%Solution Afor 6minutes.Chromatograph the Resolution solution,and record the peak responses as directed for Procedure:the relative retention times are about 1.0for clavulanic acid and 2.5for amoxicillin;the tailing factor for the clavulanic acid peak is not more than 2.0;the column efficiency determined from the clavulanic acid peak is not less than 2000theoretical plates;and the resolution,R,between the clavulanic acid peak and the amoxicillin peak is not less than 13.Chromatograph the Standard solution,and record the peak responses as directed for Procedure:the relative standard deviation for replicate injections is not more than 2%.
Procedure—
Separately inject equal volumes (about 20µL)of the Standard solutionand the Test solutioninto the chromatograph,record the chromatograms,and measure the peak responses.Calculate the percentage,in terms of clavulanate potassium equivalent,of each impurity in the Clavulanate Potassium taken by the formula:
10(237.3/205.1)(C)(ri/rS),
in which 237.3is the molecular weight of clavulanate potassium;205.1is the molecular weight of clavulanate lithium;Cis the concentration,in mg per mL,of USP Clavulanate Potassium RSin the Standard solution;riis the peak response of an individual impurity peak in the chromatogram obtained from the Test solution;and rSis the clavulanic acid peak response in the chromatogram obtained from the Standard solution.The sum of all the impurity peaks is not greater than 2%.
Assay—
pH4.4sodium phosphate buffer—
Dissolve 7.8g of monobasic sodium phosphate in 900mLof water,adjust with phosphoric acid or 10Nsodium hydroxide to a pHof 4.4±0.1,dilute with water to make 1000mL,and mix.
Mobile phase—
Prepare a suitable mixture of pH4.4sodium phosphate bufferand methanol (95:5),and pass through a membrane filter having a 0.5-µm or finer porosity.Make adjustments if necessary (see System Suitabilityunder Chromatography á621ñ).
Standard preparation—
Dissolve an accurately weighed quantity of USP Clavulanate in water to obtain a solution having a known concentration of about 0.25mg per mL.
Assay preparation—
Transfer about 50mg of Clavulanate Potassium,accurately weighed,to a 200-mLvolumetric flask,dissolve in and dilute with water to volume,and mix.
Resolution solution—
Dissolve a suitable quantity of amoxicillin in Standard preparationto obtain a solution containing about 0.5mg of amoxicillin and 0.25mg of clavulanate lithium per mL.
Chromatographic system(see Chromatography á621ñ)—
The liquid chromatograph is equipped with a 220-nm detector and a 4-mm ×30-cm column that contains 3-to 10-µm packing L1.The flow rate is about 2mLper minute.Chromatograph the Standard preparation,and record the peak responses as directed for Procedure:the column efficiency determined from the analyte peak is not less than 550theoretical plates;the tailing factor for the analyte peak is not more than 1.5;and the relative standard deviation for replicate injections is not more than 2.0%.Chromatograph the Resolution solution,and record the peak responses as directed for Procedure:the relative retention times are about 0.5for clavulanic acid and 1.0for amoxicillin;and the resolution,R,between the amoxicillin and clavulanic acid peaks is not less than 3.5.
Procedure—
Separately inject equal volumes (about 20µL)of the Standard preparationand the Assay preparationinto the chromatograph,record the chromatograms,and measure the responses for the major peaks.Calculate the quantity,in µg,of clavulanic acid (C8H9NO5)in each mg of the Clavulanate Potassium taken by the formula:
(200CP/W)(rU/rS),
in which Cis the concentration,in mg per mL,of USP Clavulanate Lithium RSin the Standard preparation;Pis the designated potency,in µg of clavulanic acid per mg,of USP Clavulanate Lithium RS;Wis the quantity,in mg,of Clavulanate Potassium taken to prepare the Assay preparation;and rUand rSare the peak responses obtained from the Assay preparationand the Standard preparation,respectively.
Auxiliary Information—
Staff Liaison:William W.Wright,Ph.D.,Scientific Fellow
Expert Committee:(PA7)Pharmaceutical Analysis 7
USP28–NF23Page 488
Pharmacopeial Forum:Volume No.29(3)Page 617
Phone Number:1-301-816-8335