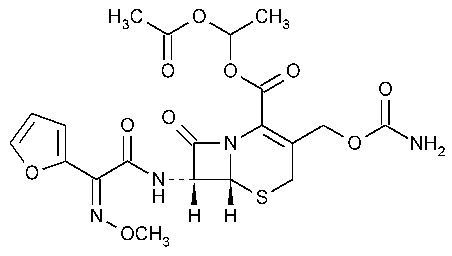
Cefuroxime Axetil
5-Thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid,3-[[(aminocarbonyl)oxy]methyl]-7-[[2-furanyl(methoxy imino)acetyl]amino]-8-oxo-,1-(acetyloxy)ethyl ester,[6R-[6a7b(Z)]]-.
(RS)-1-Hydroxyethyl (6R,7R)-7-[2-(2-furyl)glyoxylamido]-3-(hydroxymethyl)-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene 2-carboxylate,72-(Z)-(O-methyloxime),1-acetate 3-carbamate [64544-07-6].
Cefuroxime Axetil is a mixture of the diastereoisomers of cefuroxime axetil (C20H22N4O10S).It contains the equivalent of not less than 745µg and not more than 875µg of cefuroxime (C16H16N4O8S)per mg,calculated on the anhydrous basis.
Packaging and storage—
Preserve in tight containers.
Labeling—
Label it to indicate whether it is amorphous or crystalline.
Identification,Infrared Absorption á197Kñ.
Crystallinity á695ñ—
Particles that do not show birefringence or exhibit extinction positions are amorphous,and particles that show birefringence and exhibit extinction positions are crystalline.
Water,Method Iá921ñ:
not more than 1.5%.
Diastereoisomer ratio—
0.2M Monobasic ammonium phosphate,Mobile phase,Internal standard solution,Resolution solution,Standard preparation,Assay preparation,and Chromatographic system—
Prepare as directed in the Assay.
Procedure—
Proceed as directed for Procedure in the Assay.Calculate the ratio of cefuroxime axetil diastereoisomer Ato the sum of the cefuroxime axetil diastereoisomers Aand Btaken by the formula:
r/(rA+rB),
in which rAand rBare the peak responses of the cefuroxime axetil diastereoisomers Aand B,respectively:between 0.48and 0.55is obtained.
Assay—
0.2M Monobasic ammonium phosphate—
Dissolve 23.0g of monobasic ammonium phosphate in water to obtain 1000mLof solution.
Mobile phase—
Prepare a suitable filtered and degassed mixture of 0.2M Monobasic ammonium phosphateand methanol (620:380).Make adjustments if necessary (see System Suitability under Chromatography á621ñ).
Internal standard solution—
Prepare a solution of acetanilide in methanol containing 5.4mg per mL.
Resolution solution—
In a 50-mLvolumetric flask,mix 10.0mLof a solution of USP Cefuroxime Axetil RSin methanol containing 1.2mg per mL,5.0mLof Internal standard solution,and 3.8mLof a solution of USP Cefuroxime Axetil Delta-3Isomers RSin methanol containing 0.16mg per mL.Dilute with 0.2M Monobasic ammonium phosphate to volume,and mix.
Standard preparation—
Transfer about 30mg of USP Cefuroxime Axetil RS,accurately weighed,to a 25-mLvolumetric flask,dissolve in methanol,dilute with methanol to volume,and mix.Promptly transfer 10.0mLof this solution to a 50-mLvolumetric flask,add 5.0mLof Internal standard solutionand 3.8mLof methanol,dilute with 0.2M Monobasic ammonium phosphateto volume,and mix.[NOTE—Use this Standard preparation promptly,or refrigerate and use on the day prepared.]
Assay preparation—
Transfer about 30mg of Cefuroxime Axetil to a 25-mLvolumetric flask,dissolve in methanol,dilute with methanol to volume,and mix.Promptly transfer 10.0mLof this solution to a 50-mLvolumetric flask,add 5.0mLof Internal standard solutionand 3.8mLof methanol,dilute with 0.2M Monobasic ammonium phosphateto volume,and mix.[NOTE—Use this Assay preparationpromptly,or refrigerate and use on the day prepared.]
Chromatographic system (see Chromatography á621ñ)—
The liquid chromatograph is equipped with a 278-nm detector and a 4.6-mm ×25-cm column containing 5-µm packing L13.The flow rate is about 1.5mLper minute.Chromatograph the Resolution solution,and record the peak responses as directed for Procedure:the relative retention times are about 0.4for acetanilide,0.8for cefuroxime axetil diastereoisomer B,0.9for cefuroxime axetil diastereoisomer A,and 1.0for cefuroxime axetil delta-3isomers;the resolution,R,between cefuroxime axetil diastereoisomer Aand Bis not less than 1.5;and the resolution,R,between cefuroxime axetil diastereoisomer Aand cefuroxime axetil delta-3isomers is not less than 1.5.Chromatograph the Standard preparation,and record the peak responses as directed for Procedure:the column efficiency is not less than 3000theoretical plates when measured using the cefuroxime axetil diastereoisomer Apeak;and the relative standard deviation for replicate injections is not more than 2.0%.
Procedure—
Separately inject equal volumes (about 10µL)of the Standard preparation and the Assay preparationinto the chromatograph,record the chromatograms,and measure the responses for the major peaks.Calculate the quantity,in µg,of cefuroxime (C16H16N4O8S)in each mg of Cefuroxime Axetil taken by the formula:
(WS/WU)(PS/100)(100–K)(RU/RS),
in which WSis the weight,in mg,of USP Cefuroxime Axetil RStaken to prepare the Standard preparation;WUis the weight,in mg,of Cefuroxime Axetil taken to prepare the Assay preparation;PSis the designated cefuroxime (C16H16N4O8S)content,in µg per mg,of anhydrous USP Ceforoxime Axetil RS;Kis the percentage water content of USP Cefuroxime Axetil RSand RUand RSare the ratios of the sum of the peak responses of the cefuroxime axetil diastereoisomers Aand Bto the peak response of the internal standard obtained from the Assay preparation and the Standard preparation,respectively.
Auxiliary Information—
Staff Liaison:William W.Wright,Ph.D.,Scientific Fellow
Expert Committee:(PA7)Pharmaceutical Analysis 7
USP28–NF23Page 407
Phone Number:1-301-816-8335