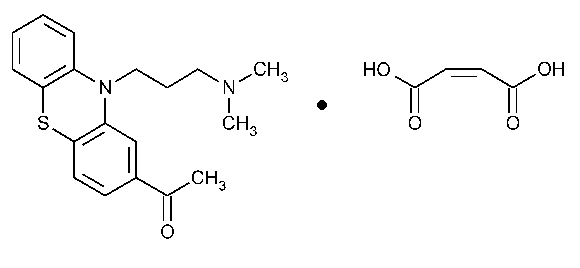
Acepromazine Maleate
Ethanone,1-[10-[3-(dimethylamino)propyl]-10H-phenothiazin-2-yl]-,(Z)-2-butenedioate (1:1).
10-[3-(Dimethylamino)propyl]phenothiazin-2-yl methyl ketone maleate (1:1) [3598-37-6].
»Acepromazine Maleate contains not less than 98.0percent and not more than 101.0percent of C19H22N2OS·C4H4O4,calculated on the anhydrous basis.
Packaging and storage—
Preserve in well-closed containers,protected from light.Store at room temperature.
Labeling—
Label it to indicate that it is for veterinary use only.
USP Reference standards á11ñ—
USP Acepromazine Maleate RS.
NOTE—Throughout the following procedures,protect test or assay specimens,the USP Reference Standard,and solutions containing them,by conducting the procedures without delay,under subdued light,or using low-actinic glassware.
Identification—
A:
Infrared Absorption á197Kñ.
B:
The retention time of the major peak for acepromazine in the chromatogram of the Assay preparationcorresponds to that in the chromatogram of the Standard preparation,as obtained in the Assay.
pHá791ñ:
between 4.0and 5.5,in a solution (1in 100).
Water,Method Iá921ñ:
not more than 1.0%.
Residue on ignition á281ñ:
not more than 0.2%.
Related compounds—
[NOTE—Conduct this test without exposure to daylight,and with the minimum necessary exposure to artificial light.]Prepare a test solution of it in a mixture of methanol and diethylamine (19:1)containing 20.0mg per mL.Prepare a Standard solution containing 0.1mg per mLby quantitatively diluting 1volume of the test solution with 200volumes of the same solvent mixture.Apply separately 10-µLportions of the test solution and the Standard solution to the starting line of a suitable thin-layer chromatographic plate (see Chromatography á621ñ)coated with a 0.25-mm layer of chromatographic silica gel mixture.Develop the chromatogram in a chromatographic chamber with a solvent system consisting of a mixture of n-heptane,isobutyl alcohol,and diethylamine (75:17:8)until the solvent front has moved about three-fourths of the length of the plate.Remove the plate from the chamber,and allow it to air-dry.Examine the plate under short-wavelength UVlight:no spot,other than the principal acepromazine spot and any at the origin,observed in the chromatogram obtained from the test solution is more intense than the principal spot observed in the chromatogram obtained from the Standard solution (0.5%).
Assay—
Mobile phase—
Add 6mLof triethylamine to 700mLof water,and adjust with phosphoric acid to a pHof 2.5.Mix 700mLof this solution and 300mLof acetonitrile,and degas.Make adjustments if necessary (see System Suitabilityunder Chromatography á621ñ).
Standard preparation—
Transfer about 50mg of USP Acepromazine Maleate RS,accurately weighed,to a 50-mLvolumetric flask,dissolve in 0.05Nhydrochloric acid,dilute with 0.05Nhydrochloric acid to volume,and mix.Transfer 5.0mLof this solution to a second 50-mLvolumetric flask,dilute with water to volume,and mix.This solution contains about 0.1mg of USP Acepromazine Maleate RSper mL.
Assay preparation—
Transfer about 50mg of Acepromazine Maleate,accurately weighed,to a 50-mLvolumetric flask;dissolve in 0.05Nhydrochloric acid;dilute with 0.05Nhydrochloric acid to volume;and mix.Transfer 5.0mLof this solution to a second 50-mLvolumetric flask,dilute with water to volume,and mix.
Chromatographic system(see Chromatography á621ñ)—
The liquid chromatograph is equipped with a 280-nm detector and a 4-mm ×15-cm column that contains 5-µm packing L7.The flow rate is about 1mLper minute.Chromatograph the Standard preparation,and record the responses as directed for Procedure:the column efficiency is not less than 1500theoretical plates;the tailing factor is not more than 2.5;and the relative standard deviation for replicate injections is not more than 2.0%.
Procedure—
Separately inject equal volumes (about 10µL)of the Standard preparationand the Assay preparationinto the chromatograph,record the chromatograms,and measure the areas for the major peaks.Calculate the quantity,in mg,of acepromazine maleate (C19H22N2OS·C4H4O4)in the portion of Acepromazine Maleate taken by the formula:
500C(rU/rS),
in which Cis the concentration,in mg per mL,of USP Acepromazine Maleate RStaken in the Standard preparation;and rUand rSare the acepromazine peak areas obtained from the Assay preparationand the Standard preparation,respectively.
Auxiliary Information—
Staff Liaison:Ian DeVeau,Ph.D.,Senior Scientist
Expert Committee:(VET)Veterinary Drugs
USP28–NF23Page 15
Pharmacopeial Forum:Volume No.29(6)Page 1832
Phone Number:1-301-816-8178