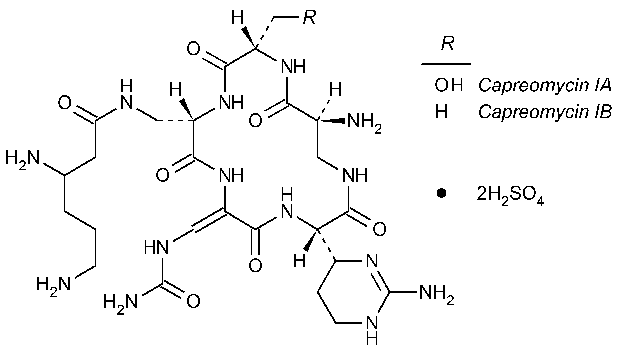
Capreomycin Sulfate
»Capreomycin Sulfate is the disulfate salt of capreomycin,a polypeptide mixture produced by the growth of Streptomyces capreolus,suitable for parenteral use.It has a potency equivalent to not less than 700µg and not more than 1050µg of capreomycin per mg.
Packaging and storage—
Preserve in tight containers.
Labeling—
Where it is intended for use in preparing injectable dosage forms,the label states that it is sterile or must be subjected to further processing during the preparation of injectable dosage forms.
Identification—
It responds to the tests for Sulfate á191ñ.
pHá791ñ:
between 4.5and 7.5,in a solution containing 30mg per mL.
Loss on drying á731ñ—
Dry about 100mg in vacuum at a pressure not exceeding 5mm of mercury at 100 for 4hours:it loses not more than 10.0%of its weight.
for 4hours:it loses not more than 10.0%of its weight.
Residue on ignition á281ñ:
not more than 3.0%,the charred residue being moistened with 2mLof nitric acid and 5drops of sulfuric acid.
Heavy metals,Method IIá231ñ:
0.003%.
Capreomycin Icontent—
Mobile phase—
Dissolve 0.5g of ammonium bisulfate in 1000mLof water,and filter through a filter having a porosity of 0.5µm or less.Prepare a mixture of this solution and methanol (550:450),and degas.Make adjustments if necessary (see System Suitabilityunder Chromatography á621ñ).
Resolution solution—
Prepare a solution of USP Capreomycin Sulfate RSin water containing about 0.25mg per mL.
Test preparation—
Prepare a solution of Capreomycin Sulfate in water containing about 0.25mg per mL.
Chromatographic system
(see Chromatography á621ñ)—The liquid chromatograph is equipped with a 268-nm detector and a 4.6-mm ×15-cm column that contains packing L10with a 3.5%carbon loading.The flow rate is about 1.5mLper minute.Chromatograph the Resolution solution,and record the peak responses as directed for Procedure:the tailing factors for the major peaks (capreomycin IAand capreomycin IB)are not more than 2.5,and the resolution between the capreomycin IApeak and the capreomycin IBpeak is not less than 1.5.
Procedure—
[NOTE—Use peak areas where peak responses are indicated.]Inject about 20µLof the Test preparationinto the chromatograph,record the chromatogram,and measure the responses for all of the peaks.Calculate the percentage of capreomycin Iin the Capreomycin Sulfate taken by the formula:
100(rIA+rIB)/rt,
in which rIAand rIBare the responses of the capreomycin IApeak and the capreomycin IBpeak,respectively,and rtis the total of the responses for all of the peaks in the chromatogram.The capreomycin Icontent is not less than 90.0%.
Other requirements—
Where the label states that Capreomycin Sulfate is sterile,it meets the requirements under Injections á1ñ,and it meets the requirements for Bacterial endotoxinsunder Capreomycin for Injection.Where the label states that Capreomycin Sulfate must be subjected to further processing during the preparation of injectable dosage forms,it meets the requirements for Bacterial endotoxinsunder Capreomycin for Injection.
Assay—
Proceed with Capreomycin Sulfate as directed under Antibiotics—Microbial Assays á81ñ.
Auxiliary Information—
Staff Liaison:William W.Wright,Ph.D.,Scientific Fellow
Expert Committee:(PA7)Pharmaceutical Analysis 7
USP28–NF23Page 335
Phone Number:1-301-816-8335